Natural Health Solutions
- The Well (Home Page)
- Water Cures Blog
- How To Do Water Cures
- What is Water Cures?
- Electrolytes & Salt
- Drinking Water
- Hire a Hydration Coach
- Acid Reflux
- Addiction Treatment
- Allergy Relief
- Blood
- Bones and Joints
- BP / Heart / Vascular
- Brain / Head / Mental
- Cancer
- Chronic Fatigue
- Cramps No More
- Dehydration
- Edema
- Fibromyalgia
- HIV / AIDS
- Immune System
- Lungs / Respiratory / Cough
- Lupus
- Lyme Disease
- Multiple Sclerosis
- Pain
- Skin Care
- Urinary Tract Infection
- Womens Health
- Water & Weight Loss
- Student Grades
- Survivalist Health Care
- Pet Water Cures
- Hydration Coach
- Testimonials
- My Water Cures
- FAQ
- In The News
- Volunteer
- About / Contact
- Red Light Therapy
- Live Blood Microscopy
What Is An Electrolyte
Lets consider the question, 'What is an Electrolyte' from a more basic viewpoint.
Electrolytes provide for electrical charges that make nerve transmission possible. Electrolytes also are involved in acid / base balance, something our bodies are constantly trying to maintain.
As described on the electrolyte directory page, an analogy of what electrolytes do in the body could be an automobile electric system. While most cars run on gasoline, without the electric system to provide the spark, they will have no way to get the gas ignited. The electric systems in the human body function with the help of chemical substances that allow the transmission of the electric impulses. These chemical substances are called electrolytes. In the human body, there are seven particular types of electrolytes that are important to our health.
Definition of Electrolytes
According to the American Heritage Dictionary, electrolyte |iˈlektrəˌlīt| is a noun.....
It is a a liquid or gel that contains ions and can be decomposed by electrolysis.
In human physiology, electrolytes are the ionized or ionizable constituents of a living cell, blood, or other organic matter.
Origins: mid 19th cent.: from electro- + Greek lutos ‘released’ (from luein ‘loosen’).
Consider what this means in the science.
Here are two ways electrolytes work. Consider this from a chemistry viewpoint.
Allowing the Splitting of Chemicals
Fluid decomposition by electrolysis requires electrolytes.
Pure Water Does Not Conduct Electricity
Most people may not be aware but pure water is a poor conductor of electricity. When there is an electrolyte added to the water, it more readily conducts electricity. When a salt, such as baking soda (sodium bicarbonate) or even table salt (sodium chloride) is added, the electricity can pass through the water. When direct current (DC) electricity is passed through the water, it causes the water to split apart into oxygen and hydrogen.
So the electrolyte provided the ions that dissolved in the water that helped the water to split apart once the electricity was available.
Electrolytes Allow for the Production of Energy
Remember, the definition above, electrolytes are the ionized or ionizable constituents of a live cell. Ionizable means that an atom or group of atoms can be electrically charged.Your car battery also uses an electrolyte. In this case, the electrolyte allows the electricity to pass to the positive terminal.
In the human body, electrolytes have a similar function.
Like your car battery, electrolytes make possible for the cells to generate energy.
A number of functions in the human body require electricity. Brain impulses, the heart pacemaker has an electric conduction and even the flexion and relaxation the muscles.
Also similar to the batter, electrolytes play a role in the control of the acidity of the body.
What Are the Electrolytes?
There are seven major or main electrolytes important to our body.
• Sodium (Na+)
• Chloride (Cl-)
• Potassium (K+)
• Magnesium (Mg++)
• Calcium (Ca++)
• Phosphate (HPO4–)
• Bicarbonate (HCO3-)What is an Electrolyte to Our Cells
Our cells use electrolytes to generate energy. They also help with the stability of cell walls.
Electrolytes are involved in the generation of electricity, help with the contraction of muscles and move fluids and water inn and out of cells. Electrolytes once inside our cells within our body, they are involved in a number of additional activities.
Hormones control the concentration of electrolytes in our body.
Specialized sensors in our kidneys monitor how much potassium, sodium and water are maintained in our bloodstream.
To maintain the levels of electrolytes, our bodies can shift water from the place of lest need to the area of greatest need. When our bodies are in a state of drought or if we get an excess of any electrolyte and not enough water is coming in to dilute and expel the excess, then a surplus of electrolyte can result.
What the Supplement Companies Don't Want You to Know
Consider This: Since our body does not process the supplement the same way as food, you can develop a surplus. Think of this as an overdose. If you overdose on some electrolytes you can have severe health consequences.If you overdose on potassium, you may get a tingling sensation in your hands and feet. You may experience muscular weakness and even temporary loss of muscle function ( called paralysis).
Even worse, abnormal heart rhythm (called cardiac arrhythmias) which can result in cardiac arrest.
Too much magnesium can cause a fall in blood pressure (called hypotension). Then lethargy, confusion, disturbances in normal cardiac heart beat (called cardiac rhythm) and eventually deterioration of kidney function, are related to severe hypotension. At its most severe, too much magnesium supplementation (called hypermagnesemia) could result in cardiac arrest.
Too much calcium (hypercalcemia) in the past resulted from the practice of using calcium supplements (1.5 to 16.5 g/day). Most often the overdose results from the combination of calcium carbonate (antacids) along with milk when used to treat stomach ulcers.
Symptoms may include confusion, delirium, coma, and if not taken care of, death.
Additionally, increased calcium supplement intake has been associated with increased risk of prostate cancer. It was found they had a risk of an advanced prostate cancer that was three times higher than men whose calcium intake was less than 500 mg/day. There was also a four times greater risk of the cancer metastasizing or spreading to other parts of the body.
In another study, smokers had an increase risk if they took in 1000 mg of calcium supplements.
But, not all studies showed the connection between calcium intake and prostate cancer. A 2006 study found that taking calcium did not increase prostate cancer risk (59. Severi G, English DR, Hopper JL, Giles GG. Re: Prospective studies of dairy product and calcium intakes and prostate cancer risk: a meta-analysis. J Natl Cancer Inst. 2006; 98(11):794-795; author reply 795.).
We suggest that the hydration status, more specifically, the intake of pure water could play a role in the outcomes. Water could be so important that it could possibly be the one factor that changed the outcomes.
Electrolytes Definition: Final Thoughts
We look at electrolytes slightly different than most. While there are elements that say reduce salt, we encourage taking more salt and along with it, more water. We suggest taking less salt than what is found in a saline IV such as you would get in a hospital.
When taking electrolytes, only naturally occurring electrolytes are safe. Any modified, chemically similar or man made electrolytes will cause harm to the body. Only naturally occurring electrolytes are safe and will produce the desired health outcomes.
More Information on Electrolytes
Recent Articles
-
Dark Screen Microscopy: What it is, does and what it can tell you.
Jul 07, 19 05:23 PM
Dark Screen Microscopy allows for a BioNutritional Auditi to look at your blood in its whole, live state -
About Water Cures and how water can help improve your health and life
Oct 25, 18 09:38 PM
About water cures: information on the Water Cures Protocol, Dr. Batmanghelidj and the founder of this site, Bob Butts.. -
Water Cures Testimonials
Apr 16, 18 09:26 PM
Water cures testimonials of those who improved their health using the water cures protocol.
October 22, 2018...
Robert Butts, founder of WaterCures.org passed away at the age of 83. He will be missed.

 |
 |
Warning: Research shows calcium supplements may be harmful to your health.
| Fixing Blood Disorders
Posted December 2016 |
 |
© 2016 Jonathan Steele
It is amazing how so many things that are new to us were common knowledge in the past. One basic practice of the Water Cures protocol is to drink at least 30-45 minutes before and 2 1/2 hours after a meal. This is not a new thought however. Notice what a magazine from 1925 had to say...
"Drink plenty of water two hours after each meal; drink none just before eating; and a small quantity if any at meal time. Do not take a bath until two hours after eating a meal, nor closer than one hour before eating. Drink a full glass of water both before and after the bath." (Golden Age, Sept. 9, 1925, pp. 784-785)
 |
New in 2016
Cancer 1, Natural Cures 0
The War...is against...
Cancer Natural Cures
---------------------
| Hand Joint Pain: How Can I Make It Go Away? |
 |
 |
to try a no cost, scientifically proven way of improving your well being?
Click Here to try the 3 Minute Challenge
Find them right here!
 Shoulder Joint Pain Relief Shoulder Joint Pain Relief |
 |
 |
What Your Doctor's Not Telling You!
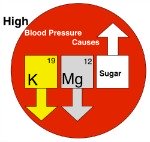 High Blood Pressure Causes High Blood Pressure Causes |
 |
 |
We started expanding on the causes...
The weirdest...chimney sweep cancer. You won't believe where it strikes.
Our theory on...
Healthy Hydration for Athletics & the reason for hydration guideline failures in the past.
We speak to organizations small and large, private and corporate.
Our scientifically proven training works to improve performance and decrease lost days due to illness.
We are currently speaking to hospitals to train staff in ways to decrease the readmission rates in several disease processes that pose high risk of <30 day readmission.
We can cut the rates by up to 70%. Ask us how.
Nurse Jon for more information on hydrating to improve performance.
Here is the Science
We are not promoting increased salt intake. We are suggesting taking salt in amounts appropriate to your bodies needs, based on water needs. Our needs are not one size fits all.
Note: Do you have CHF or Kidney Disease? Then.....
THIS IS NOT FOR YOU.
 |
Read This Study
And Decide for Yourself
 |
For Most of Us, No!
According to a JAMA study.
A Review of the above JAMA Article
Study: Salt May Not Be All Bad?
Listen to your body. Do not use this if you are under a doctors care. Do not stop taking medications without consulting your doctor. If you are on medications, consult your doctor if you start the Water Cures Protocol as it may change your needs.
 |
Click Here to Discover How
 |
Click Here
Sign Up for your biweekly newsletter.
 |
 |
 |
If you are looking for an affordable alternative natural health care for depression, consider the Water Cures protocol.
Like What You're Learning? Please share your likes on Facebook
 |
 |
 |
 |
Hi, my name is Sharon. The webmaster of this site is my nurse, my personal water coach and my husband.
As this site was first being built, I had a headache and as usual took an Ibuprofen. Impatient for it to start working, I decided to try the Water Cures. I took a pinch of salt and a glass of water. Then I took a second pinch of salt and another glass of water. My headache was gone in less than 5 minutes.
From my personal experience, it usually takes 30 to 45 minutes for Ibuprofen to work. Some have found it takes ibuprofen 24 minutes to start working.
Yet on the Water Cures protocol, my headache was gone in 5 minutes.
Its simple: give your body what it needs and your body will give you what you need, the ability to feel great.
Water Cures was the solution for the elimination of my headache. It is what I will use from now on.
Why not give it a try yourself.
